More than half of the aquaculture species produced are fed\r\nformulated feeds, and several studies have focused on additional terrestrial\r\nproteins as ingredient sources – mainly legumes like field...
More than half of the aquaculture species produced are fed\r\nformulated feeds, and several studies have focused on additional terrestrial\r\nproteins as ingredient sources – mainly legumes like field peas, lupin and fava\r\nbeans – that are regionally and organically produced. Legumes provide nitrogen\r\nfor themselves and subsequent plants and thus reduce the overall fertilization\r\nneeds during crop rotation.
Several different cultivars of lupin have been tested with\r\ndifferent aquaculture species and with generally promising results. Lupin\r\ndigestibility has been shown to exceed that of soy in Atlantic salmon. Extruded\r\nlupin seed meal has been found to offer good digestibility coefficients,\r\nespecially for protein and energy, in trout and turbot, and can greatly\r\nsubstitute fishmeal in European seabass diets. At least 40 percent of fishmeal\r\nprotein in diets of the black tiger shrimp (Penaeus monodon) can be replaced by\r\nlupin kernel meal (used on a protein‐equivalent basis), without adverse impacts\r\non growth. And a study showed that the Andean lupin (Lupinus mutabilis) can\r\nreplace at least half of the fishmeal protein – equivalent to a third of the\r\ntotal protein – in the diet of Pacific white shrimp (Litopenaeus\r\nvannamei) without negatively influencing growth and feed conversion. However,\r\ninformation about the effects of metabolic and immune parameters is missing so\r\nfar.
This article – adapted and summarized from the original\r\npublication [M. Weiss et al. 2020. Lupin kernel meal as fishmeal\r\nreplacement in formulated feeds for the Whiteleg Shrimp (Litopenaeus\r\nvannamei). Aquaculture Nutrition 2020;00:1–11.] – reports on a study\r\nto evaluate narrowleaf lupin (Lupinus angustifolius) seed meal as a sustainable\r\ndiet component for Pacific white shrimp in controlled feeding experiments conducted\r\nin a recirculating aquaculture system.
This study is part of the project “TRansition paths to\r\nsUstainable legume‐based systems in Europe” (TRUE), and it has received funding\r\nfrom the European Union’s Horizon 2020 research and innovation programme under\r\ngrant agreement no. 727973. The authors thank the staff from the “Centre for\r\nAquaculture Research” (ZAF) for conducting water quality analyses and daily\r\nroutine activities.
Study setup
The study was conducted in a recirculating aquaculture\r\nsystem (RAS) system at the Center for Aquaculture Research, Alfred Wegener\r\nInstitute in Bremerhaven, Germany. The setup included 18 separate tanks with a\r\nwater treatment system including a mechanical filter, a protein skimmer, a\r\nbiofilter and ozone treatment. During the trial, physical water parameters were\r\nmeasured every day (91.93 ± 4.95 percent for dissolved oxygen; 7.53 ± 0.11 for\r\npH; 26.11 ± 0.69 degrees-C for temperature and 15.96 ± 0.53 grams per L for\r\nsalinity). Twice a week, the concentrations of nitrogen compounds were\r\ndetermined, with mean concentrations of 0.27 ± 0.245 mg/L for ammonium, 2.25 ±\r\n3.692 mg/L for nitrate and 122.71 ± 96.16 mg/L for nitrite.
L. vannamei postlarvae (PL13, mean weight ~ 3 mg ± 0.5\r\nmg, mean ± standard deviation) were obtained from a commercial shrimp hatchery\r\nin Florida (USA) and grown for seven weeks. Tanks were each stocked with 25\r\nindividuals at a mean biomass per unit of 90.22 ± 0.86 grams. Controlled\r\nfeeding was maintained for eight weeks and treatments were done in\r\nquadruplicate. Weight and length gains were recorded at the beginning, and\r\nafter four and eight weeks. At the end of the experiment, haemolymph samples\r\nwere collected for further analyses, after determining the molting stage of\r\neach animal and excluding freshly molted animals due to known impacts of\r\nmolting on various metabolic and immune parameters.
Four experimental, isonitrogenous and isocaloric, diets were\r\nformulated to meet the requirements of L. vannamei during the study,\r\nconsidering energy content, protein and amino acid profile, lipid and fatty\r\nacid composition, vitamins and minerals. As necessary, single amino acids\r\n(methionine and lysine) were added to balance the amino acid profile. The diets\r\nincluded a control with fishmeal as main protein source; diets L10 and L20 with\r\n10 and 20 percent, respectively, lupin kernel meal replacing fishmeal; and L30\r\nwith 30 percent lupin kernel meal in complete replacement of fishmeal. A\r\ncommercial shrimp diet (Beeskow, Germany; protein 390 grams per kg, lipid 90\r\ngrams per kg, ash 90 grams per kg, fiber 15 grams per kg) without lupin meal\r\nwas tested in duplicate for comparison.
For detailed information on the experimental system and\r\ndiets; phenoloxidase activity; total and differential haemocyte counts; and\r\nstatistical analyses, refer to the original publication.
Results and discussion
All animals accepted the feeds and showed acceptable growth.\r\nThe average survival rate of shrimp across all lupin meal supplemented diets\r\nwas 68.3 ± 7.3 percent. For the animals receiving the control diet, it was 63.0\r\n± 5.0 percent, and survival rates were not statistically different between\r\ntreatments. After eight weeks, the body weight of the animals differed\r\nsignificantly depending on the diet (Table 1). The animals fed the control\r\nfeed, the L10 feed and the commercial diet were the heaviest. Shrimp fed the\r\nL20 diet were significantly lighter than control and L10 diet but did not\r\ndiffer from the commercial diet. Shrimp fed the L30 diets had significantly\r\nlower body weights than all other treatments. These findings are also reflected\r\nin the specific growth rate, which is above 1.5 for all diets but drops below 1.0\r\nin shrimp fed the L30 diet. The reduced growth trend in the L30 treatment was\r\nalready evident after four weeks.
Weiss, Lupin meal,
Table 1
| \r\n Diet \r\n | \r\n \r\n Average biomass,\r\n start (g) \r\n | \r\n \r\n Average biomass,\r\n end (g) \r\n | \r\n \r\n Weight gain (g) \r\n | \r\n \r\n Specific growth\r\n rate (% BW/day)* \r\n | \r\n
| \r\n Com \r\n | \r\n \r\n 3.60 ± 0.00 \r\n | \r\n \r\n 9.50 ± 0.55 \r\n | \r\n \r\n 5.898 \r\n | \r\n \r\n 1.731 \r\n | \r\n
| \r\n Control \r\n | \r\n \r\n 3.62 ± 0.05 \r\n | \r\n \r\n 10.37 ± 0.55 \r\n | \r\n \r\n 6.747 \r\n | \r\n \r\n 1.878 \r\n | \r\n
| \r\n L10 \r\n | \r\n \r\n 3.61 ± 0.05 \r\n | \r\n \r\n 9.66 ± 0.58 \r\n | \r\n \r\n 6.044 \r\n | \r\n \r\n 1.756 \r\n | \r\n
| \r\n L20 \r\n | \r\n \r\n 3.61 ± 0.02 \r\n | \r\n \r\n 8.53 ± 0.55 \r\n | \r\n \r\n 4.924 \r\n | \r\n \r\n 1.537 \r\n | \r\n
| \r\n L30 \r\n | \r\n \r\n 3.60 ± 0.03 \r\n | \r\n \r\n 6.24 ± 0.61 \r\n | \r\n \r\n 2.644 \r\n | \r\n \r\n 0.984 \r\n | \r\n
\r\nTable 1. Growth results of the feeding experiments for initial weight, weight\r\ngain and specific growth rate of L. vannamei. Replicate numbers were 24, 63,\r\n73, 73 and 59 for Com, control, L10, L20 and L30, respectively. *Percent body\r\nweight per day.
The haemolymph phenoloxidase (PO) activity [an important component\r\nof the shrimp immune defense] was highest in animals fed the L10 diet and\r\nsignificantly lower when fed the commercial diet (Fig. 1a). Activity in the\r\ncontrol and the L20 and L30 diets was intermediate, without significant\r\ndifferences compared with the commercial (Com) or the L10 diet.
The glucose level was significantly higher in L10 fed\r\nanimals (28.36 ± 6.44 mg/dl) than in L30 fed animals (19.71 ± 2.73 mg/dl) (Fig.\r\n1b). Glucose levels of shrimp fed the commercial feed (27.0 ± 4.15 mg/dl), the\r\ncontrol feed (22.89 ± 2.26 mg/dl) and L20 (24.73 ± 5.0 mg/dl) were intermediate\r\nand did not differ significantly from L10 or L30.
The mean total haemocyte count (THC) values were 277.6 ±\r\n118.7 ×105 cells ml/L. Total haemocytes were higher when shrimp were fed low\r\nlevels of lupin (L10) than the THC values of shrimp fed the commercial and the\r\ncontrol feed. THC values gradually decline with higher lupin inclusion levels.\r\nNo significant differences between the dietary treatments occurred. However, we\r\nobserved a tendency that semi-granular cells are promoted in animals fed the\r\nL10 diet but are reduced with further increasing lupin content in the feed\r\n(Fig. 1c).
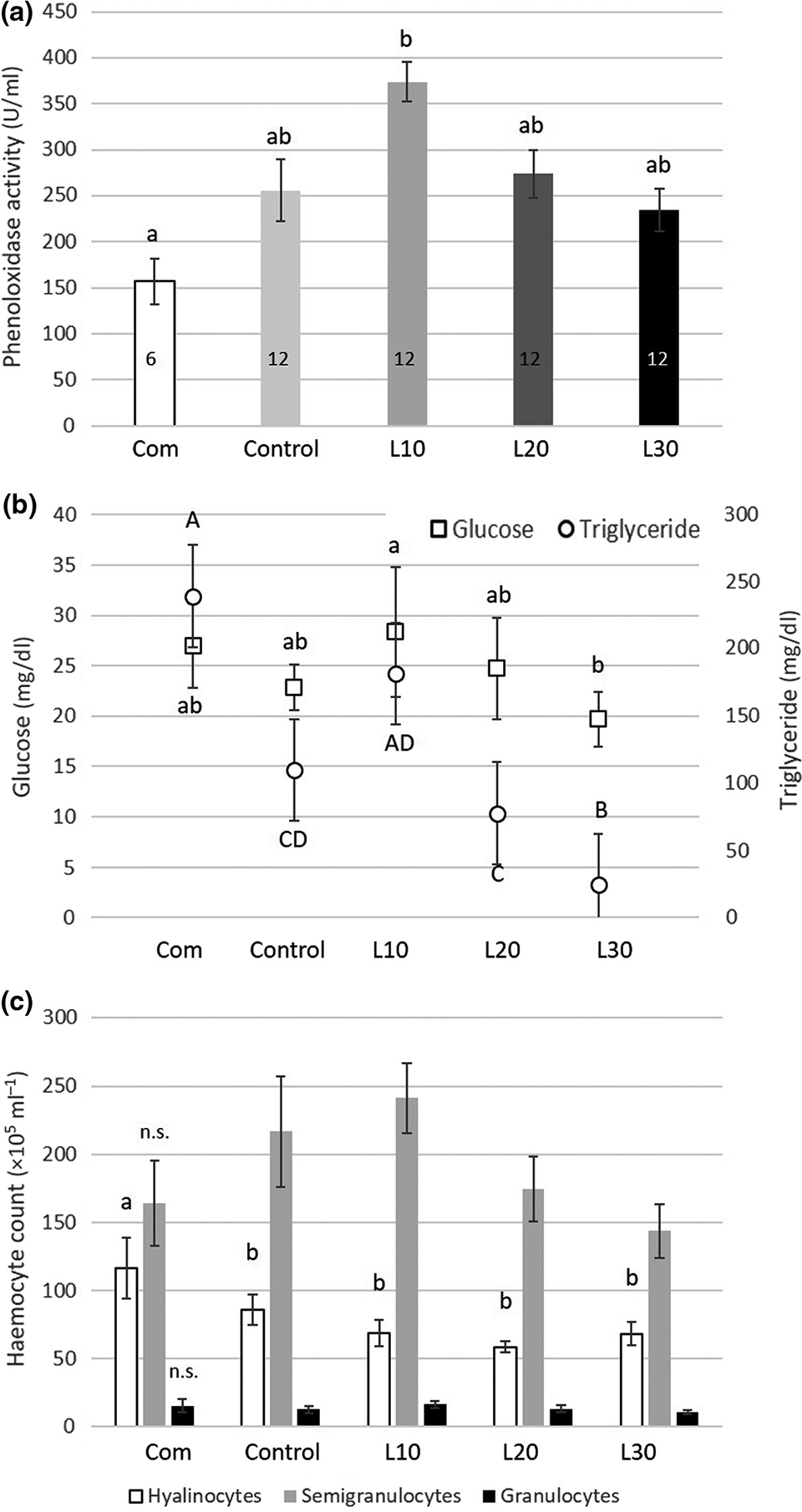
Fig. 1: Results of haemolymph analyses. Com: commercial\r\nfeed; Control: control feed; L10: 10 percent of the feed is lupin meal; L20: 20\r\npercent of the feed is lupin meal; L30: 30 percent of the feed is lupin meal.\r\nSignificant differences are indicated by different letters. Replicate numbers\r\nare indicated in the bars for a), for b) and c) is Com = 6 individuals (ind.),\r\nControl, L10, L20 and L30 = 12 ind., each ind. measured in 3 technical\r\nreplicates. (a) Phenoloxidase activity in shrimp haemolymph given as mean ± SE.\r\nData were transformed to reach normality. (b) Glucose and acylglyceride levels\r\nmeasured in shrimp haemolymph given as mean ± SD. (c) Differential haemocyte\r\ncount.
Sustainable and viable alternative proteins remain high\r\npriorities for future aquaculture development. The results of our study clearly\r\nindicate the applicability of lupin seed meal as a replacement for fishmeal in\r\ndiets for L. vannamei culture. Inclusion is, as with many terrestrial\r\nalternative sources, recommendable but only within clear limitations. The\r\ngrowth of L. vannamei indicates that increasing inclusion rates of\r\nlupin meal exceeding 100 g/kg (replacing 40 percent of fishmeal) in the feed\r\ncause a progressive decline in shrimp performance.
New ingredients for aquafeeds, especially plant‐derived\r\nproducts, can have impacts on the metabolism of the animal that might not be\r\nexpressed on the growth level but in metabolic parameters. The metabolic data\r\nfrom our study show that while inclusion of 10 percent lupin meal has no\r\nnegative influence, increasing supplementation with lupin meal (20 and 30\r\npercent) progressively deteriorates the physiological status of L.\r\nvannamei shrimp towards lower metabolite content in the total haemolymph.
In general, total protein levels in the haemolymph of shrimps\r\nfed the different diets are in the range reported for animals maintained under\r\nsimilar conditions. Our results show low but stable haemolymph protein levels\r\nfor all diets, indicating a limited but sufficient protein supply not affected\r\nby the rate of lupin meal inclusion in the diets tested.
Our results also provide evidence that the inclusion of\r\nlupin meal in the feed has a modulating effect on the shrimp’s immune system\r\nwith positive enhancement of the haemocytes and the phenoloxidase (PO) system\r\nwhen the lupin is included in moderate levels (10 percent; Fig. 1a,c). Most of\r\nthe designed diets resulted in slightly higher PO activity, but the L10 diet‐treated\r\nanimals showed a significant increase in PO activity compared with the\r\ncommercial diet. Higher values of PO activity were also found by various other\r\nauthors in L. vannamei when other immune‐stimulating and probiotic\r\ningredients were included in the feed.
Perspectives
Our results demonstrate the successful inclusion of dehulled\r\nlupin seed meal in feeds for L. vannamei without adverse effects on\r\nsurvival, growth performance or metabolic parameters for inclusion rates of up\r\nto 100 grams per kg of feed. High inclusion rates (300 grams per kg lupin seed\r\nmeal) resulted in reduced growth performance and nutritional status. An immune‐stimulating\r\neffect on the shrimp was detected for an inclusion level of 100 grams per kg\r\nlupin meal, based on an increase in phenoloxidase activity.
\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n\r\n
These results prove that dehulled lupin seed meal is a\r\nsuitable, regional alternative protein source for aquaculture feeds that can\r\nsupply good quality protein to L. vannamei shrimp and can replace\r\nsignificant amounts of diet fishmeal. For future diet developments, higher\r\nsubstitution rates might be achieved by supplementing a mix of lupin meal and\r\nother regional plants, such as fava bean. This might provide a more balanced\r\nnutritional supply and make use of the immuno‐stimulating effect of moderate\r\nlupin inclusion rates. Additionally, further research is required to assess\r\nmethods for lupin pretreatment to enhance digestibility.
Source: Global Alliance Aquaculture

Ditulis oleh
Tim Minapoli
Kontributor
Pakar di bidang akuakultur dengan pengalaman lebih dari 15 tahun. Aktif berkontribusi dalam pengembangan industri perikanan Indonesia.
